Loading blog...
Health Sector Research: Methods, Ethics, and What Makes It Different From Other Research Fields
Health research operates at the intersection of scientific rigor, ethical obligation, and often life-or-death policy stakes. That combination changes everything about how it is done.
Maya Chen
Apr 18, 2026•3 min read
Health sector research is where methodology and ethics are most tightly intertwined. The people being researched are often vulnerable: patients, caregivers, people living with chronic conditions, children, or communities experiencing disease burden. The data being collected is often sensitive. And the findings are often used to inform decisions that affect access to healthcare, resource allocation, and public health policy.
Researchers who approach health sector work with the same methodological toolkit they use for consumer or market research often find themselves unprepared for the additional obligations, processes, and design considerations that come with the territory.
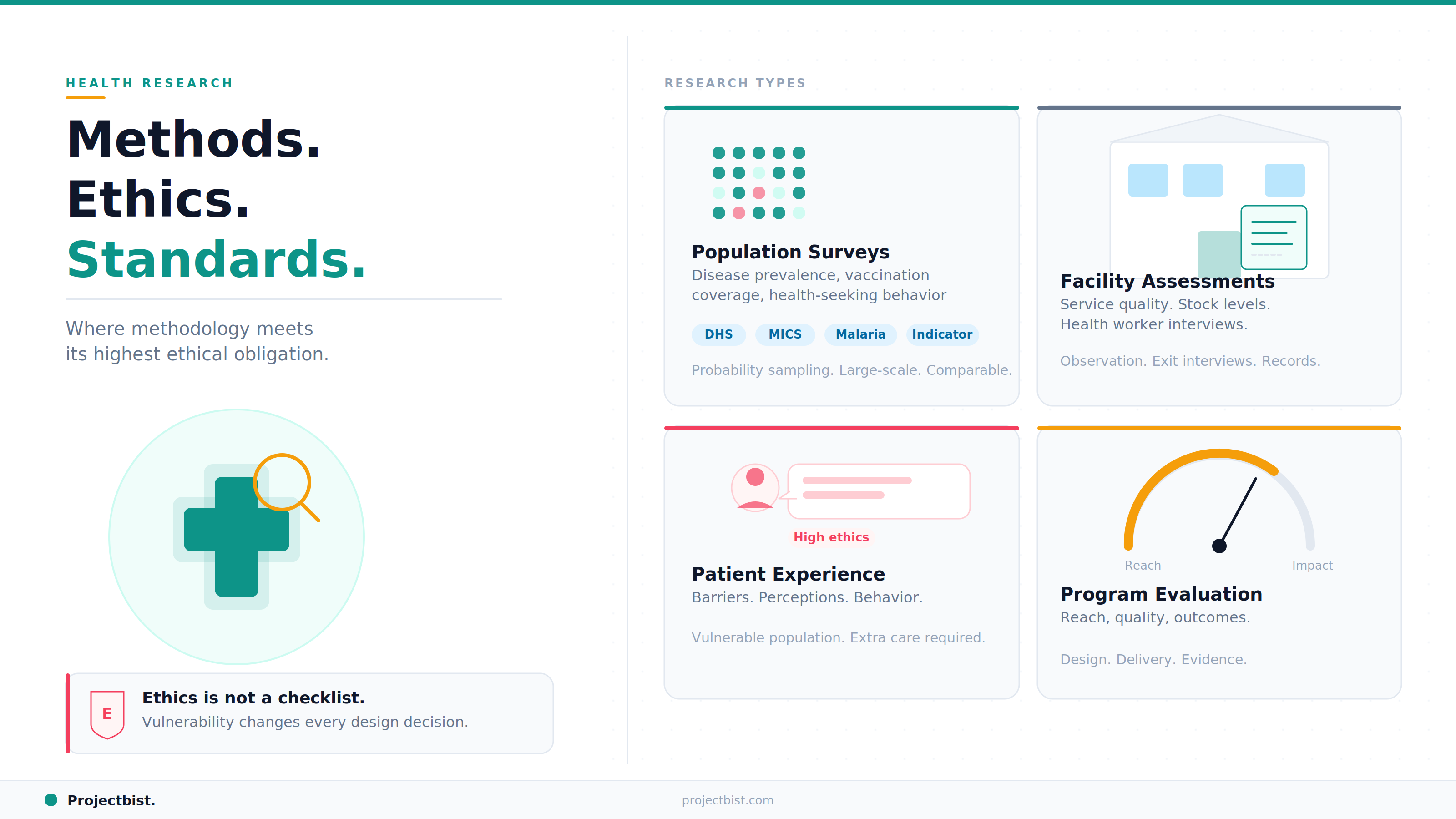
The Main Types of Health Sector Research
1. Epidemiological and Population Health Surveys
Household surveys measuring disease prevalence, health-seeking behavior, nutritional status, vaccination coverage, and health system utilization. These studies follow probability sampling principles and often require large sample sizes to produce statistically reliable estimates at disaggregated levels such as district, gender, or age group.
Well-known examples include the Demographic and Health Survey (DHS), the Multiple Indicator Cluster Survey (MICS), and Malaria Indicator Surveys, which follow standardized internationally validated protocols to allow comparability across countries and over time.
2. Health Facility Assessments
Studies evaluating the quality, availability, and readiness of healthcare services at facility level. Methods include direct observation of service delivery, client exit interviews, health worker interviews, and review of facility records and stock levels.
3. Patient and User Experience Research
Qualitative and quantitative studies exploring how patients and healthcare users experience health services: what barriers they face, what motivates or discourages healthcare-seeking, and how they perceive the quality of care they receive. These studies are methodologically similar to customer experience research but carry significantly greater ethical obligations because of the vulnerability of the research population.
4. Health Program Evaluation
Assessments of the reach, quality, and impact of health interventions: vaccination campaigns, maternal health programs, HIV treatment scale-up, nutrition interventions. These evaluations typically combine quantitative survey data on coverage and outcomes with qualitative exploration of implementation quality and contextual factors affecting outcomes.
The Ethics Layer in Health Research
All health research involving human participants requires ethics review and approval from a recognized institutional review board (IRB) or research ethics committee before data collection begins. This requirement is not optional and not simply a procedural hurdle. It exists because the potential for harm in health research is real, as established by historical cases of exploitative research that led to the Declaration of Helsinki and the Belmont Report.
For research in low- and middle-income countries, ethics review often requires both in-country ethics committee approval and, if the study is funded or led by an institution from another country, ethics approval from that institution as well. Research teams should plan for this dual-review process in their project timelines, as approvals can take weeks to months.
Sensitive Topic Research in Health Settings
Many health research topics require specific methodological adaptations to collect honest, reliable data. Topics like HIV status, sexual behavior, reproductive health, mental health, substance use, and domestic violence all require approaches that protect respondent privacy, minimize social desirability bias, and create conditions where respondents feel safe to answer honestly.
Adaptations commonly used include: audio computer-assisted self-interview (ACASI) for sensitive questions where respondent-entered data reduces interviewer influence; anonymous survey designs where no identifying information is collected; and community engagement protocols that brief local leaders and trusted figures before fieldwork begins.
In health research, you are asking people to share information that is personal, sometimes stigmatized, and often linked to real vulnerability. The methods have to respect that.
FAQ
Do all health research projects need ethics approval?
Yes. Any research involving human participants in a health context requires formal ethics review and approval before data collection begins. This applies whether the research is qualitative or quantitative, small or large scale. The only exception is research using entirely anonymized secondary data that cannot be traced back to individuals.
What is the DHS and can it be used as a secondary data source?
The Demographic and Health Survey (DHS) is a nationally representative household survey program covering health, population, and nutrition topics in over 90 countries. DHS datasets are publicly available through the DHS Program website (dhsprogram.com) for registered researchers and are widely used as secondary data sources for health and demographic analysis.
How long does ethics approval typically take for a health research project?
Timelines vary widely by country and institution. In well-resourced settings, full board review can take four to six weeks. In many low- and middle-income country research settings, review processes can take two to four months or longer. Researchers should factor this into project planning and budget timelines.
Newsletter
Get ProjectBist research notes in your inbox
Personalize your updates! Subscribe to ProjectBist's Newsletter and choose from the following categories.
Related Articles
View all
How to Manage a Multi-Country Research Project Without Losing Data Quality, Timeline, or Your Mind

Rapid Assessment Methodology: How to Design Research That Is Fast Without Being Sloppy
Systematic Reviews and Literature Synthesis: When the Data Already Exists and Your Job Is to Make Sense of It